Research |
 |
hh
Mainww |
| Press reportsww |
Peopleww |
We have designed and built a variety microchannel networks generating stable linear and exponential gradients of soluble factors, temperature and gas concentrations. The following projects are currently active. (1) Chemotaxis of D. discoideum amoebas in exponential gradients and in shallow chambers with linear gradients, in collaboration with Bill Loomis, UCSD. (2) Response of D. discoideum amoebas to sharp changes in the level of attractant, in collaboration with Rick Firtel, UCSD. (3) Chemotaxis of primary neutrophils in exponential concentration profiles. (4) Chemotropism of yeast in response to pheromone gradients, in collaboration with Andre Levchenko, Johns Hopkins University. (5) Thermotaxis of E. coli bacteria. (6) Aerotaxis of E. coli and B. subtilis bacteria.
We built a series of microfluidic devices for on-chip cultures of bacteria and yeast. The devices enable culturing cells at constant medium conditions up to very high cell densities and reaching extremely high densities, tracking a colony starting from a single cell for >10 generations at a single-cell resolution, rapidly adding and removing soluble factors without loosing cells, testing a range of medium conditions and multiple cell lines in a single experiment, controlling the oxygen tension in the medium, and temporarily immobilizing cells for high-resolution imaging. We use the devices to study the ammonium metabolism and antibiotic response in E. coli in collaboration with Terry Hwa, UCSD. In addition, our bacterial culture devices are currently used in the Andre Levchenko lab, Johns Hopkins University (simultaneous stimulation of multiple E. coli strains and the Ilka Bischofs lab, Heidelberg University (signaling in B. subtilis bacteria).
We designed and applied novel microfluidic perfusion devices for experiments on blood cells: rolling and adhesion of platelets, rolling of neutrophils, and shear stress response of endothelial cells. To seal the perfusion devices against substrates with plated cells, we developed an original magnetic clamp. Our perfusion chambers are currently used in several projects: (1) TIRF microscopy of rolling and adhesion of leukocytes to various substrates; (2) shear stress response of human endothelial cells; (3) study of the strength of adhesion of D. discoideum amoebas to different substrates, in collaboration with Bill Loomis, UCSD; (4) study of possible circadian cycles in Paramecium protozoa.
We developed a technology of high refractive index gels with physiological rigidity that enables combining the traction force and TIRF microscopy of adherent animal cells. We applied it to concurrently visualize the maps of traction forces and points of adhesion of human endothelial cells. We also developed a microfluidic technique to measure the elastic moduli of thin layers of soft gels. Our technologies are currently used in the following projects: (1) localization of various adhesion-related fluorescently labeled proteins on the cell surface adjacent to the substrate at different substrate rigidities; (2) dependence of expression of various proteins in cells plated on a substrate on the substrate rigidity; (3) concurrent TIRF and traction force microscopy on high refractive index soft substrates.
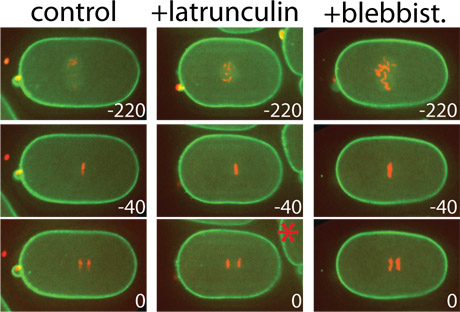
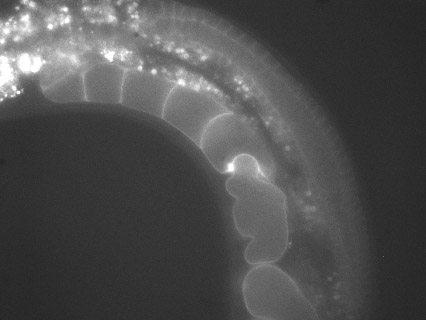
We developed a microwell device enabling the extraction of shell-free eggs from C. elegans worms and the imaging of their early stages of development with the possibility to add and remove desired soluble factors at well-defined time points. The devices are currently used by several C. elegans labs. We also developed an easy-to-use device that enables adjustable immobilization of C. elegans at different larval and adult stages for extended high-resolution imaging. The devices are now being used in the labs of Karen Oegema, UCSD, Sandra Encalada, TSRI, and Olivier Cinquin, UCI.
Lymphocyte imaging
In collaboration with Leor Weinberger, UCSF, we built several different microwell devices for fluorescence imaging of primary human lymphocytes and cultured Jurkat cells to study the development of HIV infection in a laboratory model at a single-cell resolution.Oxygen tension and hypoxia
We designed and built a computer 10-channel gas mixer, providing 10 gas mixtures with different oxygen concentration on demand. We used it with 24-well plates, custom-made multiwell plates and microfluidic devices to study various aspects of cellular behavior (gene expression, motility, viability, etc.) as functions of oxygen tension in the medium. Currently, we are applying the technique to study oxygen taxis of E. coli and testing the oxygen response of zebrafish.Kinetics and thermodynamics of protein folding
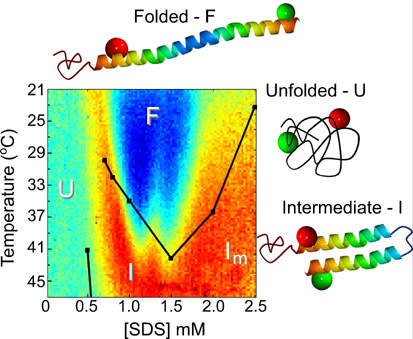
We developed and applied a series of microfluidic devices to study the kinetics of protein folding and to obtain equilibrium diagrams of protein conformations at high resolution using measurement of the Foerster resonance energy transfer (FRET) from protein molecules labeled with two fluorescent dyes. This project is a collaboration with Ashok Deniz, TSRI. In particular, we built a device with orthogonal gradients of temperature and of concentration of small molecule (SDS) to obtain a 100x100 point 2D diagram of protein conformations from a single FRET micrograph. We also used the porosity of the microfluidic him material (PDMS) to realize in-situ deoxygenation and substantially improve the dye photostability for single-molecule FRET (smFRET) experiments, making it possible to reduce the minimal sampling time to 50us. Finally, we built an optimized microfluidic mixer that improved the time resolution of smFRET folding kinetics experiments ~50-fold bringing it to 0.2 ms. We have also developed a new protein folding technique based on ultra-fast (500 ns) cooling and now finishing our first study with this technique.Biolistic delivery with a gene gun
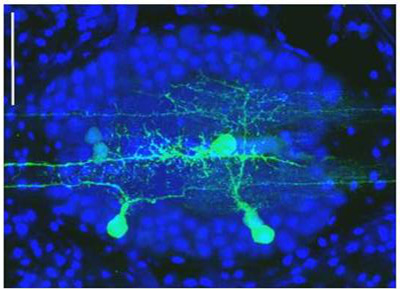
We designed and built a series of gene guns for biolistic delivery of nucleic acids and fluorescent dyes into cell in internal layers of tissues and organisms. All these guns are based on our patented technology that eliminates the gas shock wave emerging of the gun barrel, thus preventing mechanical damage to the target tissue. The most recent model of the gen gun delivers 1um gold beads to neurons in live mouse brain and 0.5um beads to adult C. elegans and neurons in brain slices. Our gene guns are presently used for different purposes in several labs: transfection and RNA-interference in leech (Hirudo medicinalis) embryos, Orit Shefi, Bar Ilan University, Israel; sparse labeling of neurons in brain slices with voltage sensitive fluorescent dyes for functional imaging.